
Technological breakthroughs
Hybrid MXene – A Breakthrough 2D Material Ushering in a New Era for High-Performance Lithium-Ion Batteries
Collected by Bảo Hiền02:49 PM @ Wednesday - 08 April, 2026
An international research team has published a comprehensive review of MXene-based hybrid composites for next-generation lithium-ion batteries (LIBs). Since its discovery in 2011, this unique class of 2D materials has gradually established itself as a “cornerstone” of the global energy storage revolution—but only when combined with suitable materials to overcome its inherent limitations.

What is MXene and why does it matter?
Amid surging global demand for energy storage—from electric vehicles and smart grids to wearable electronics—the race to develop superior electrode materials has never been more intense. MXene is emerging as one of the most promising candidates.
MXene is a family of two-dimensional (2D) transition metal carbides and nitrides, generally expressed as Mₙ₊₁XₙTₓ, where M represents a transition metal (Ti, V, Nb, etc.), X is carbon or nitrogen, and Tₓ denotes surface functional groups (–OH, –F, –O). These materials are produced by selectively etching the A-layer from MAX phases using hydrofluoric acid, molten salts, or electrochemical methods.
What makes MXene exceptional is its rare combination of properties: metallic-level electrical conductivity (up to 10,000 S/cm), hydrophilic surfaces that facilitate lithium-ion diffusion, large specific surface area, and flexible layered structures capable of accommodating charge–discharge cycles. Since its discovery, MXene research has evolved through distinct phases: 2014–2016 focused on pure synthesis; post-2017 on carbon hybridization; 2019–2021 on composites with metal oxides and sulfides; and since 2022 on integration with polymers, organic systems, and multi-component architectures.
Three core challenges and how composites address them
Despite its advantages, pristine MXene faces three major barriers in practical applications:
• Restacking: Van der Waals forces cause MXene sheets to restack, reducing surface area and blocking Li⁺ diffusion channels.
→ Solution: Introduce carbon, polymers, or metal oxides as spacers.
• Surface oxidation: Moisture and elevated temperatures degrade the structure, reducing conductivity and electrode lifespan.
→ Solution: Apply protective coatings such as g-C₃N₄ or suitable polymers.
• Limited volumetric capacity: The layered structure offers insufficient Li storage sites, resulting in lower energy density than conversion-type materials.
→ Solution: Combine with high-capacity metal oxides such as SnO₂ and Fe₂O₃.
Four major MXene composite systems and representative performance
1. MXene/Carbon nanostructures
Graphene, carbon nanotubes (CNTs), and porous carbon serve dual roles: enhancing electrical conductivity and acting as physical spacers to prevent restacking and maintain open Li⁺ channels.
• Hollow Ti₃C₂Tₓ/carbon nanofibers (electrospun): 306.5 mAh/g at 40 mA/g—far exceeding conventional MXene paste electrodes (81 mAh/g)
• V₂CTₓ/5% CNT: 446 mAh/g; V₂CTₓ/5% graphene: 460 mAh/g; Coulombic efficiency >98% after 140 cycles
• MXene@SiNPs@NC foam: 1,658 mAh/g after 100 cycles; 857 mAh/g after 500 cycles
• 3D-MCC-LFP cathode (MXene/CNT/cellulose/LiFePO₄): areal capacity of 19.2 mAh/cm²—the highest reported for LFP cathodes
Technical note: Optimal carbon content should remain below 10 wt%. Beyond this threshold, carbon may block pores and hinder Li⁺ diffusion.
2. MXene/Metal oxides
Transition metal oxides (TMOs) such as SnO₂, Fe₂O₃, and TiO₂ offer high theoretical capacities but suffer from poor conductivity and volume expansion. MXene effectively compensates for both drawbacks.
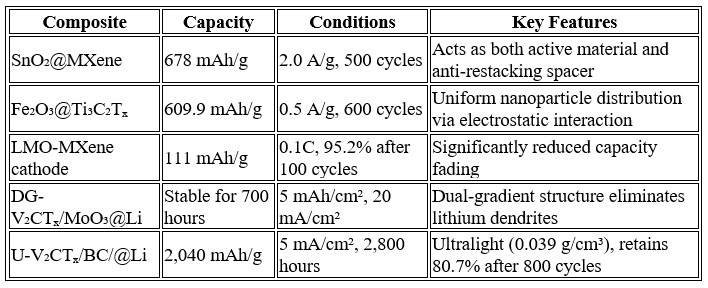
3. MXene/Polymers
Conductive polymers (e.g., polyaniline, polypyrrole) and non-conductive polymers (e.g., PVP, PVA) play complementary roles.
• PI/MXene nanofiber separator: tensile strength 19.6 MPa, electrolyte uptake 832%, initial discharge capacity 126.7 mAh/g—far superior to conventional Celgard 2400 (95.6 mAh/g)
• Solid polymer electrolyte PEO-LiTFSI/MXene (3.6 wt% MXene): ionic conductivity of 2.2×10⁻⁵ S/cm at room temperature—enabling all-solid-state batteries
• Ti₃C₂-g-P4VP-Ag composite: dendrite-free lithium deposition with Coulombic efficiency >98%
4. MXene/MoS₂ and MOFs
MoS₂ offers a high theoretical capacity of 670 mAh/g via multi-electron reactions.
• SnS₂@C/MXene: reversible capacity of 867.1 mAh/g after 100 cycles
• DFT calculations show that –O and –F surface groups reduce Li diffusion activation energy from 0.80 eV to 0.22 eV
• (CoS@NHC)@MXene aerogel: 1,145.9 mAh/g at 1 A/g after 800 cycles—demonstrating the power of hierarchical 3D porous structures
Five priority research directions
1. Interface engineering: Covalent bonding, heteroatom doping, and self-assembly to stabilize interfaces and enhance mechanical durability
2. Sustainable synthesis: Electrochemical etching, molten salt, and solvent-free methods for scalable, non-toxic production
3. Advanced architectures: 3D hierarchical, yolk–shell, and porous scaffold structures to mitigate volume expansion and improve electrolyte penetration
4. Flexible integration: Flexible batteries, wearables, and all-solid-state systems for next-generation electronics and EVs
5. In situ characterization: Synchrotron XRD, XPS, EIS, and TEM to understand degradation mechanisms and optimize material design
Source:
Umme Kalsoom et al., “MXene-based hybrid composites for lithium-ion batteries: advances in synthesis strategies and electrochemical performance,” Ionics (2025), 31:10053–10073.
Received: May 8, 2025 · Accepted: August 7, 2025 · Published: August 28, 2025.
NIFERCO Sets Target for Fifth Consecutive Year of Record Revenue
Date 08/04/2026Rising Energy Costs Reshape Caustic Soda Supply Chains in 2026
Date 07/04/2026Quantum battery that breaks the rules of charging
Date 07/04/2026

